Strong Differential Monovalent Anion Selectivity in Narrow Diameter Carbon Nanotube Porins
 ACS Nano 2020, 14, 5, 6269–6275
ACS Nano 2020, 14, 5, 6269–6275
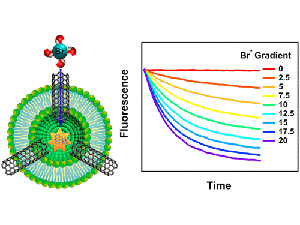
The abstract reads as follows: Inner pores of carbon nanotubes combine extremely fast water transport and ion selectivity that could potentially be useful for high-performance water desalination and separation applications. We used dye-quenching halide assays and stopped-flow spectrometry to determine intrinsic permeability of three small monovalent halide anions (chloride, bromide, iodide) and one pseudohalide anion (thiocyanate) through narrow 0.8 nm diameter carbon nanotube porins (CNTPs). These measurements revealed unexpectedly strong differential ion selectivity with permeabilities of different ions varying by up to 2 orders of magnitude. Removal of the negative charge from the nanotube entrance increased anion permeability by only a relatively small factor, indicating that electrostatic repulsion was not a major determinant of CNTP selectivity. First principle molecular dynamics simulations revealed that the origin of this strong differential ion selectivity is partial dehydration of anions upon entry into the narrow CNTP channels.
This work was supported as part of the Center for Enhanced Nanofluidic Transport (CENT), an Energy Frontier Research Center funded by the U.S. Department of Energy, Office of Science, Basic Energy Sciences under Award No. DESC0019112.
https://pubs.acs.org/doi/abs/10.1021/acsnano.0c02423
